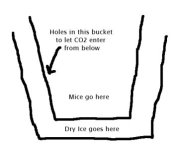
If I use dry ice to euthanize via CO2 inhalation, I usually put the mice/rats in a good-sized container that is very deep (like a bucket, garbage can, etc.) I then take the dry ice and drop it into a gladware container with holes in it and pour hot water in before putting on the lid. The CO2 comes pouring out the holes at a rapid rate so that when I place the gladware down in the bottom of the container, CO2 levels rise very rapidly.
Actually, if you let the CO2 levels rise a little more gradually, the mice lose consciousness more calmly and with less stress than dropping them into a higher concentration. They aren't feeling suffocated. The feeling of suffocation comes from the response to low Oxygen levels. You get hypoxic (low on oxygen), you struggle to breathe and you feel paniced.
CO2 can anesthetize and kill an animal without setting off the hypoxia response of being deprived of oxygen. It will stimulate them to breathe faster at lower blood levels, then once a threshhold is reached, it actually slows respirations. Higher levels do work faster, though. I have a friend who (not very brightly) stuck his head down into CO2 vapor and took one big breath...and nearly passed out on the floor. LOL, he didn't think it would work that fast. He said the world started to go black for an instant. He didn't however, feel like he was suffocating. DISCLAIMER: I DO NOT THINK ANYONE SHOULD TRY THIS FOR THEMSELVES...JUST AS I TOLD HIM HE WAS A DOOFUS TO DO SO. But, since he did pull that stunt, I thought I'd share the info from it.
CO2 does not work by displacing Oxygen in the tank. High blood CO2 levels will kill an animal regardless of their Oxygen levels. Room air contains just 21% Oxygen and 3% Carbon Dioxide. You'd probably only have to up CO2 in the air to maybe 10-15% to eventually kill an animal (although it wouldn't be as instant as the higher levels). Animals under anesthesia can easily die at just 3 times the carbon dioxide levels in their blood while breathing 100% Oxygen.